Introduction
Once again I find myself writing about a subject that is new to all of us. Now James at Practice Index does like to challenge me, so not content with the 2017-18 challenge of GDPR, he has given me the 2018-19 challenge of FMD… and I thought we were friends! The last thing we needed was yet another three-letter abbreviation (TLA) in primary care, but guess what, we got one! FMD is the Falsified Medicines Directive. And, to top it all off, it’s another EU directive, so expect it to be as clear as the GDPR! And just like the GDPR, I think there’s going to be a lot of ‘hurry up and wait’ for the General Practice Toolkit for FMD, as at the time of writing the status of the toolkit is “not yet completed”. Bringing back memories of pre-April 2018 by any chance? Let’s throw Brexit into the mix too; will it or won’t it happen? That’s a bit like the FMD – will it or won’t it? Read on to learn more.
Background
The EU Falsified Medicines Directive (2011/62/EU) , to give it its full title, was published in 2011 and was made applicable in 2013. The FMD was introduced to harmonise measures to ensure that medicines within the EU are safe and trade in medicine has effective control measures. Why? Well, falsified medicines present a threat to public health, including adverse reactions and no improvement in the patient’s condition. The overarching aim of the FMD is to prevent harm as a result of falsified medicines. Measures within the FMD include:1
- Obligatory safety features – a unique identifier and anti-tampering device on the outer packaging of medicines
- A common, EU-wide logo to identify legal online pharmacies
- Tougher rules on the import of active pharmaceutical ingredients
- Strengthened record-keeping requirements for wholesale distributors
Why does this matter?
Again, similar to GDPR, there is a time frame. Member states have until 9th February 2019 to implement the final element of the FMD, which is the Delegated Regulation (EU) 2016/161 which explains the requirements for the safety features which are to appear on the packaging of medicinal products. The two key safety features being:2
- Unique Identifier (UI) – Alphanumerical code enabling the identification and authentication of individual packs (2D barcode)
- Anti-tampering Device (ATD) – Device allowing the verification of whether a pack has been opened or tampered with
The image below illustrates the safety features: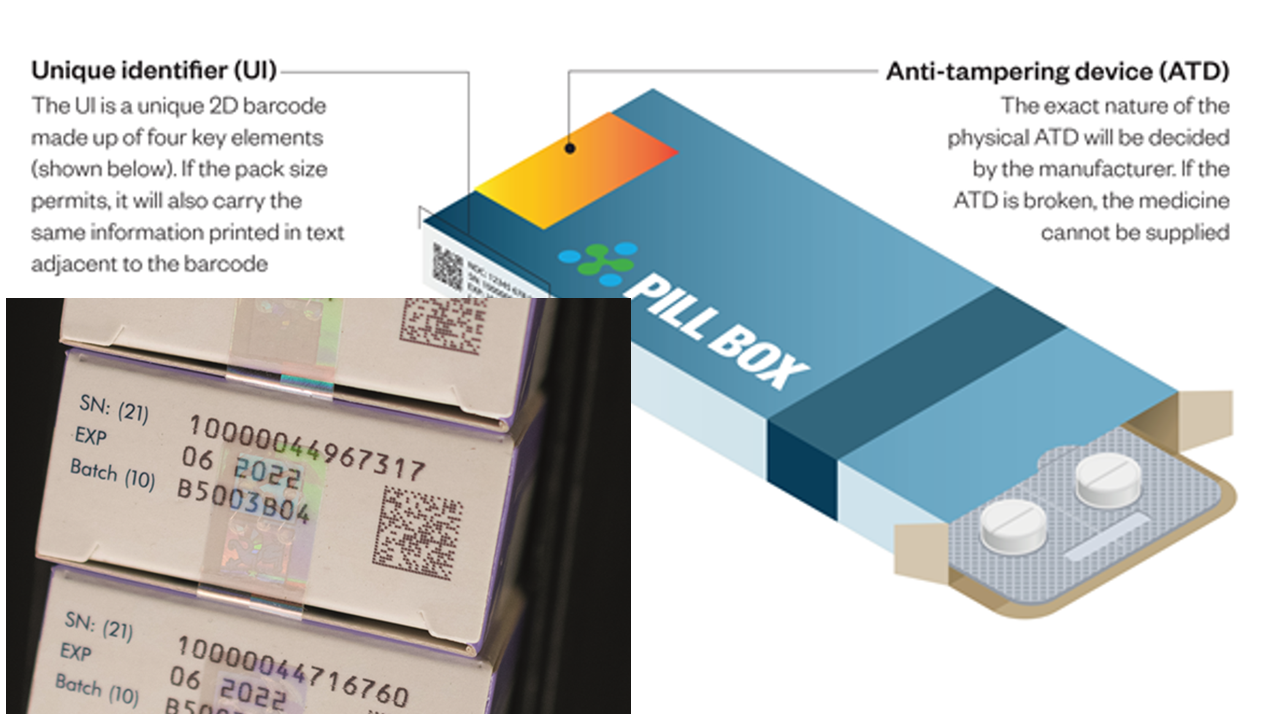
Image credit: The Pharmaceutical Journal, FMD How It Will Work
Still not clear why it applies to general practice? Well, let’s break down the overall process. Any movement of the medicine through the supply chain is referred to as verification, and when the medication is handed to the patient, this is the end of the supply chain and this is referred to as decommissioning. By decommissioning the medication, the UI is changed to reflect the fact that the pack has been supplied to the patient. This information is held in the national repository, which is run by SecurMed UK. Any other pack bearing the same UI (i.e. falsified medicine) will not or cannot be successfully decommissioned.
All practices will need to have the capability to decommission medicines; for dispensing practices, there will be a requirement to decommission packs of medication at the point of being issued to the patient. For non-dispensing practices, there’s a requirement to decommission vaccines once administered to patients.
Decommissioning is achieved by using an approved scanner which has the following decoding capabilities:
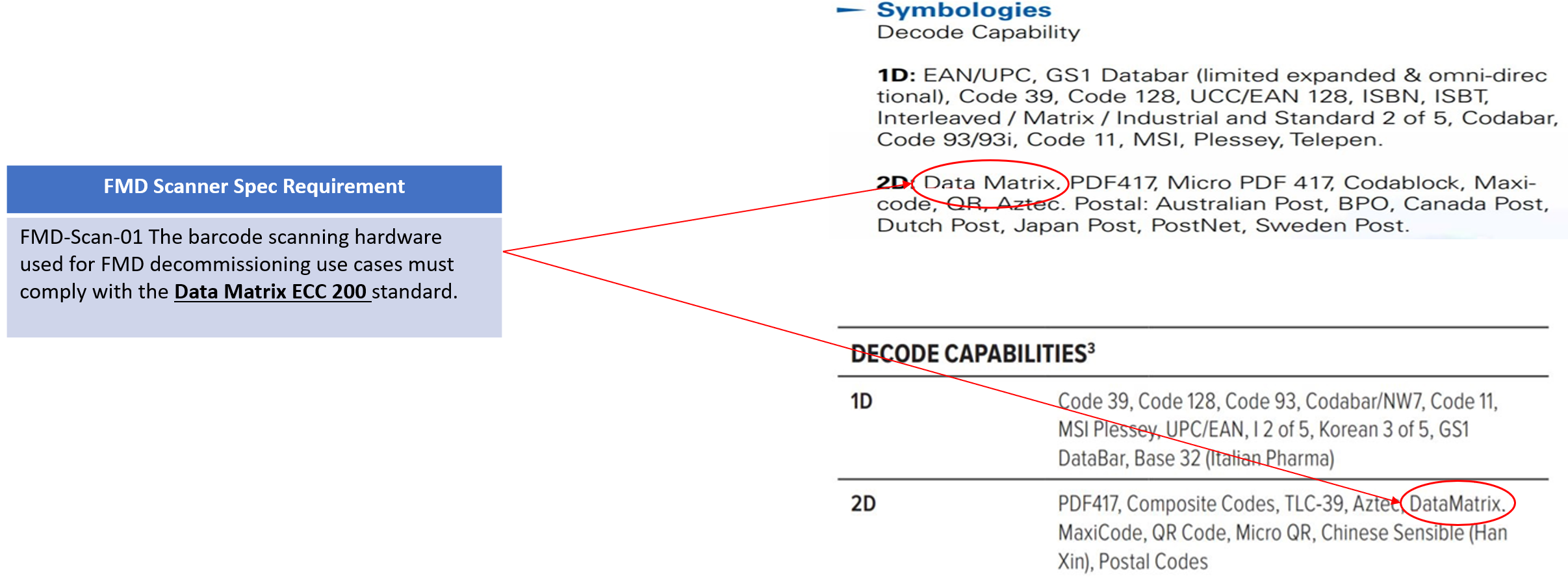
Image credit: developer.nhs.uk, FMD Implementation Toolkits
Brexit and the FMD
For over two years Brexit has been in the news and this week there has been intense scrutiny of Brexit and the actual impact on the UK if we leave the EU. Now I’m not a politician and will therefore keep my political views out of this article; otherwise I may find myself taking a no-deal exit from Practice Index! But according to an article in the BMA, if we leave the EU under a no-deal Brexit, “it is unlikely that the Directive would come into effect in its current form”, but given the need to ensure patient safety and to maintain and access safe medicines, an effective relationship must be maintained with the EU. So, for now, it’s over to Teresa May to see if she can deliver what the nation wants.
Keep an eye on the relevant page on the BMA website for updates as Brexit takes its course – https://www.bma.org.uk/advice/employment/gp-practices/service-provision/falsified-medicines-directive
Implications
What are the implications? Well, time is going to be an issue (as if there wasn’t enough to do already!). Regardless of your dispensing status, the FMD will mean that staff will have to decommission vaccines and medication at the point of administration or issue. Sadly, the workload for dispensing practices will increase and this could mean practices will need additional resources… and will have to fund said resources.
All practices are going to need hardware and software, and the BMA will continue to work with the DoH and NHS(E), and have argued strongly that the latter must fund the equipment required to enable practices to comply with the FMD.
Conclusion
Just like the GDPR, FMD will happen (he says, with Brexit up in the air!) and we have an implementation date of the 9th February 2019 to ensure that we’re prepared to comply with the Directive. Unfortunately, general practice is once again reliant on the wider NHS to provide the necessary guidance that practices need to prepare and subsequently implement the FMD.
Extra reading!
See this free factsheet for the key points.
———-
Topics trending in the forum:
Patient querying historic entry
HR conundrum no 5: Unpaid parental leave…
Advice please – resignation
Bank Holiday calculator 2019-2020





January 18, 2019 at 11:51 am
This is very useful, are any practices actually doing anything in preparation as yet?